Reprogram Biosciences
Commercialization Strategy for mRNA-based Solid Tumor Therapeutics
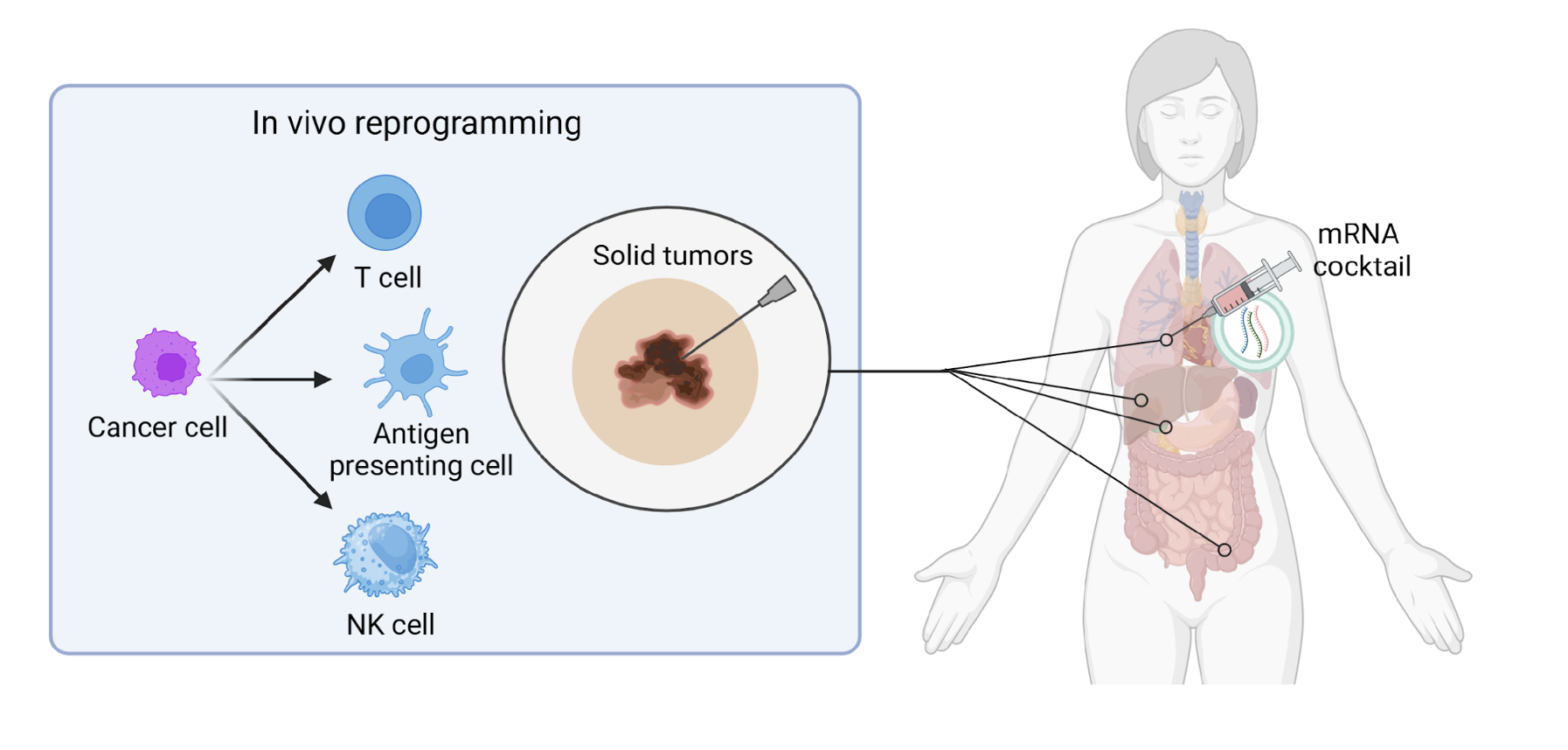
Project Overview
Reprogram Biosciences is a biotech startup developing innovative mRNA-based therapeutics for solid tumor treatment. As a member of the commercialization strategy team, I conducted in-depth market research and developed strategic recommendations to position their therapeutic platform within the competitive oncology landscape. I work alongside with a cross-functional team of medical students/PhDs, MBA candidates, and the company founder to integrate clinical insights into business strategy.
Key Challenge
How to effectively position a novel mRNA therapeutic platform in a crowded oncology market while identifying the most promising indication areas and go-to-market strategies.
Methodology & Approach
- Market Landscape Analysis: Conducted extensive research on the $150B+ global oncology market, analyzing competitor positioning, treatment paradigms, and unmet medical needs
- Indication Prioritization: Developed a systematic framework to evaluate potential tumor indications based on market size, competitive landscape, and technical feasibility
- Stakeholder Mapping: Identified and analyzed key stakeholders including KOLs, regulatory bodies, payers, and potential strategic partners
- Competitive Intelligence: Deep-dive analysis of 10+ competing platforms in the mRNA therapeutics space
Key Findings & Insights
- Identified that clinical skepticism and lack of clear biomarker data are slowing the adoption of novel mRNA-based reprogramming therapies for solid tumors.
- Uncovered high-potential white space in TNBC, ovarian, and NSCLC indications for personalized mRNA therapeutics, where emerging biomarker targeting strategies remain underleveraged in current therapeutic pipelines.
- Found that mRNA-based immune reprogramming is most commercially viable when positioned as a complement to existing checkpoint inhibitors. This led to a recommendation to pursue co-development or partnership opportunities with immuno-oncology players for expanded clinical trials.
- Mapped the value chain of clinical translation and identified regulatory, manufacturing, and IP challenges unique to personalized mRNA therapies. Proposed a phased market entry model starting with orphan indications and modular platform demonstrations.
Impact & Results
The strategic recommendations directly influenced the company's R&D prioritization, and market research framework was adopted for ongoing competitive intelligence and strategic planning.
Tools & Technologies Used
Key Learnings
This project deepened my understanding of the biotech commercialization landscape and the critical importance of market-driven product development. I learned to balance scientific innovation with commercial viability, and developed expertise in navigating complex regulatory environments.